Endometriosis: what’s the evidence for this enigmatic disease?
Posted on 2nd June 2014 by Antonio Simone Lagana

To date endometriosis early diagnosis and management seems to be far from reaching a final shape. Despite the large number of published papers, the causes of the disease are still unclear: accumulating evidence [1] suggests that gene mutations could play a role to alter the correct process of female reproductive tract organogenesis. In this condition, it seems that the primordial and undifferentiated cells which will form the different part of the female reproductive tract could remain misplaced during the organogenesis. During the postpubertal age, these cells could differentiate under the influence of hormonal changes, and form the typical endometriotic implants in the peritoneal cavity. Once established, the endometriotic foci have a dynamic interplay with the immune system: this last seems to address the inflammatory response of the peritoneal microenviroment.
Regardless of new evidence, the symptoms and signs of the disease are better known: while some women with endometriosis can experience painful symptoms and/or infertility, others have no symptoms at all. The exact prevalence of endometriosis is unknown but estimates range from 2 to 10% of women of reproductive age, to 50% of infertile women [2, 3].
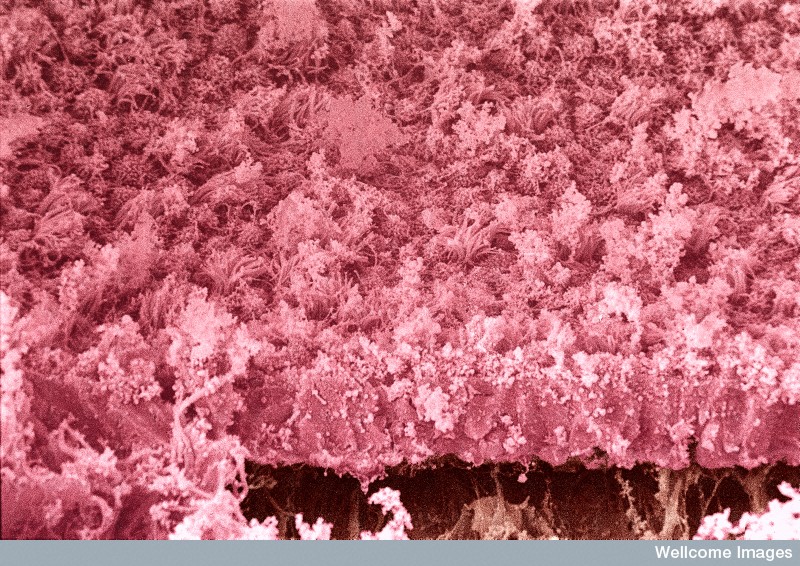
Endometriosis on an ovarian cyst. Colour-enhanced, scanning electron micrograph of endometriosis. A layer of endometrial epithelium (pink) can be seen growing on an ovarian cyst (brown). Ciliated and secretory cells are visible as well as considerable fibrin debris. Yorgos Nikas, Wellcome Images
A new update [4] from the European Society of Human Reproduction and Embriology (ESHRE) tried to shed light about this extremely important topic, with a new set of recommendations. In particular, this guideline provides 83 recommendations on diagnosis of endometriosis and on the treatment of endometriosis-associated pain and infertility, on the management of women in whom the disease is found incidentally (without pain or infertility), on prevention of recurrence of disease and/or painful symptoms, on treatment of menopausal symptoms in patients with a history of endometriosis and on the possible association of endometriosis and malignant tumors growth. The guideline development was initiated by members of the 2005 guideline development group (GDG), supplemented with advice from additional experts in the field. If you are interested, you can find the full text of this paper here.
In this commentary, we will try to shed light to the most important recommendations stated by the GDG.
Diagnosis
Clinicians should consider the diagnosis of endometriosis:
- In the presence of gynaecological symptoms such as: dysmenorrhoea, non-cyclical pelvic pain, deep dyspareunia, infertility and fatigue in the presence of any of the above.
- In women of reproductive age with non-gynaecological cyclical symptoms (dyschezia, dysuria, haematuria and rectal bleeding, shoulder pain).
Clinicians may consider the diagnosis of:
- Deep endometriosis in women with (painful) induration and/or nodules of the rectovaginal wall found during clinical examination or visible vaginal nodules in the posterior vaginal fornix.
- Ovarian endometrioma in women with adnexal masses detected during clinical examination.
- Endometriosis in women suspected of the disease even if the clinical examination is normal.
It is mandatory to:
- Perform a laparoscopy to diagnose endometriosis, although evidence is lacking that a positive laparoscopy ‘without histology’ proves the presence of disease.
- Confirm a positive laparoscopy by histology, since positive histology confirms the diagnosis of endometriosis even though negative histology does not exclude it.
- Obtain tissue for histology in women undergoing surgery for ovarian endometrioma and/or deep infiltrating disease, to exclude rare instances of malignancy.
About imaging, the GDG recommended:
- To perform TVS to diagnose or to exclude an ovarian endometrioma.
- That clinicians base the diagnosis of ovarian endometrioma in premenopausal women on the following ultrasound characteristics: ground glass echogenicity and one to four compartments and no papillary structures with detectable blood flow.
Moreover, clinicians should be aware that the usefulness of:
- 3D ultrasound to diagnose rectovaginal endometriosis is not well established.
- Magnetic Resonance Imaging (MRI) to diagnose peritoneal endometriosis is not well established.
- Finally, it is mandatory to assess ureter, bladder and bowel involvement by additional imaging if there is a suspicion based on history or physical examination of deep endometriosis, in preparation for further management.
Regarding biomarkers, clinicians are recommended not to use them in endometrial tissue, menstrual or uterine fluids and/or immunological biomarkers, including CA-125, in plasma, urine or serum to diagnose endometriosis.

Action figure series, Zhang Haiying, 2009. neutralnotes.tumblr.com
Medical treatment of endometriosis-associated pain
The GDG recommends clinicians to:
- Counsel women with symptoms presumed to be due to endometriosis thoroughly, and to empirically treat them with adequate analgesia, combined hormonal contraceptives or progestagens.
- Prescribe hormonal treatment (hormonal contraceptives, progestagens, anti-progestagens, or GnRH agonists as one of the options, as it reduces endometriosis-associated pain).
- Take patient preferences, side effects, efficacy, costs and availability into consideration when choosing hormonal treatment for endometriosis-associated pain.
Clinicians can consider:
- Prescribing a combined hormonal contraceptive, as it reduces endometriosis-associated dyspareunia, dysmenorrhea and non-menstrual pain.
- The continuous use of a combined oral contraceptive pill in women suffering from endometriosis-associated dysmenorrhea.
- The use of a vaginal contraceptive ring or a transdermal (oestrogen/progestin) patch to reduce endometriosis-associated dysmenorrhoea, dyspareunia and chronic pelvic pain.
Regarding progestagens and anti-progestagens, the GDG recommends:
- To use progestagens or anti-progestagens as one of the options, to reduce endometriosis-associated pain.
- That clinicians take the different side-effect profiles of progestagens and anti-progestagens into account when prescribing these drugs, especially irreversible side effects (e.g. thrombosis and androgenic side effects).
- To consider prescribing a levonorgestrel-releasing intrauterine system (LNG-IUS) as one of the options to reduce endometriosis-associated pain.
Regarding GnRH agonists, the GDG recommends:
- To use them as one of the options for reducing endometriosis-associated pain, although evidence is limited regarding dosage or duration of treatment.
- To prescribe hormonal add-back therapy to coincide with the start of GnRH agonist therapy, to prevent bone loss and hypoestrogenic symptoms during treatment.
- To give careful consideration to the use of GnRH agonists in young women and adolescents, since these women may not have reached maximum bone density.
Moreover:
- In women with pain from rectovaginal endometriosis, refractory to other medical or surgical treatment, clinicians can consider prescribing aromatase inhibitors in combination with oral contraceptive pills, progestagens or GnRH analogues, as they reduce endometriosis-associated pain.
- Clinicians should consider non-steroidal anti-inflammatory drugs (NSAIDs) or other analgesics to reduce endometriosis-associated pain.
Surgical treatment of endometriosis-associated pain
When endometriosis is identified at laparoscopy, clinicians are recommended to surgically treat endometriosis, as this is effective for reducing endometriosis-associated pain, i.e. ‘see and treat’.
In this condition, clinicians may consider both ablation and excision of peritoneal endometriosis to reduce endometriosis-associated pain.
When performing surgery in women with ovarian endometrioma, clinicians should perform cystectomy instead of drainage and coagulation, as cystectomy reduces endometriosis-associated pain. Clinicians can consider performing cystectomy rather than CO2 laser vaporization in women with ovarian endometrioma, because of a lower recurrence rate of the endometrioma.
Clinicians can consider performing surgical removal of deep endometriosis, as it reduces endometriosis-associated pain and improves quality of life. The GDG recommends that clinicians refer women with suspected or diagnosed deep endometriosis to a center of expertise that offers all available treatments in a multidisciplinary context.
As a radical solution, clinicians may consider hysterectomy with removal of the ovaries and all visible endometriosis lesions in women who have completed their family and failed to respond to more conservative treatments. Women should be informed that hysterectomy will not necessarily cure the symptoms or the disease.
Clinicians should not perform laparoscopic uterosacral nerve ablation (LUNA) as an additional procedure to conservative surgery to reduce endometriosis-associated pain. Conversely, clinicians should be aware that presacral neurectomy (PSN) is effective as an additional procedure to conservative surgery to reduce endometriosis-associated midline pain, but it requires a high degree of skill and is a potentially hazardous procedure.
Clinicians can use oxidised regenerated cellulose during operative laparoscopy for endometriosis, as it prevents adhesion formation. Conversely, it is not reasonable for clinicians to use icodextrin after operative laparoscopy for endometriosis to prevent adhesion formation, as no benefit has been shown. Finally, clinicians should be aware that other anti-adhesion agents (polytetrafluoroethylene surgical membrane, hyaluronic acid products) have been studied and proven effective for adhesion prevention in the context of pelvic surgery, although not specifically in women with endometriosis.
Regarding adjunctive hormonal treatment, clinicians should not prescribe:
- Preoperative hormonal treatment to improve the outcome of surgery for pain in women with endometriosis.
- Adjunctive hormonal treatment in women with endometriosis for endometriosis-associated pain after surgery, as it does not improve the outcome of surgery for pain.
About secondary prevention of disease and painful symptoms:
- The GDG states that there is a role for prevention of recurrence of disease and painful symptoms in women surgically treated for endometriosis. The choice of intervention depends on patient preferences, costs, availability and side effects.
- In women operated on for an endometrioma (≥3 cm), clinicians should perform ovarian cystectomy, instead of drainage and electrocoagulation, for the secondary prevention of endometriosis-associated dysmenorrhoea, dyspareunia and non-menstrual pelvic pain.
- After cystectomy for ovarian endometrioma in women not immediately seeking conception, clinicians are recommended to prescribe combined hormonal contraceptives for the secondary prevention of endometrioma.
- In women operated on for endometriosis, clinicians are recommended to prescribe post-operative use of a LNG-IUS or a combined hormonal contraceptive for at least 18 – 24 months, as one of the options for the secondary prevention of endometriosis-associated dysmenorrhoea, but not for non-menstrual pelvic pain or dyspareunia.
Finally, the GDG does not recommend the use of nutritional supplements, complementary or alternative medicine in the treatment of endometriosis-associated pain, because the potential benefits and/or harms are unclear. However, the GDG acknowledges that some women who seek complementary and alternative medicine may feel benefit from this.

Parachute. Kyung, December 2013. endometriosisartjourney.tumblr.com
Treatment of endometriosis-associated infertility
In infertile women with endometriosis, clinicians should not prescribe hormonal treatment for suppression of ovarian function to improve fertility.
In infertile women with AFS/ASRM Stage I/II endometriosis, clinicians:
- Should perform operative laparoscopy (excision or ablation of the endometriosis lesions) including adhesiolysis, rather than performing diagnostic laparoscopy only, to increase ongoing pregnancy rates.
- May consider CO2 laser vaporization of endometriosis, instead of monopolar electrocoagulation, since laser vaporization is associated with higher cumulative spontaneous pregnancy rates.
In infertile women with ovarian endometrioma undergoing surgery, clinicians should perform excision of the endometrioma capsule, instead of drainage and electrocoagulation of the endometrioma wall, to increase spontaneous pregnancy rates. The GDG recommends that clinicians counsel women with endometrioma regarding the risks of reduced ovarian function after surgery and the possible loss of the ovary. The decision to proceed with surgery should be considered carefully if the woman has had previous ovarian surgery.
In infertile women with AFS/ASRM Stage III/IV endometriosis, clinicians can consider operative laparoscopy, instead of expectant management, to increase spontaneous pregnancy rates.
the GDG recommends clinicians not to prescribe adjunctive hormonal treatment before or after surgery to improve spontaneous pregnancy rates, as suitable evidence is lacking.
Finally, no evidence was found showing a beneficial effect of different types of nutritional supplements, complementary and alternative treatments for improving infertility in women with endometriosis.
Assisted Reproduction Technology (ART) in women with endometriosis
In infertile women with AFS/ASRM Stage I/II endometriosis, clinicians may perform Intrauterine insemination with controlled ovarian stimulation, instead of expectant management, as it increases live birth rates.
The GDG recommends the use of ART for infertility associated with endometriosis, especially if tubal function is compromised or if there is male factor infertility, and/or other treatments have failed. This even after surgery, since cumulative endometriosis recurrence rates are not increased after controlled ovarian stimulation.
Moreover, clinicians can prescribe GnRH agonists for a period of 3 – 6 months prior to B treatment with ART to improve clinical pregnancy rates in infertile women with endometriosis.
In infertile women with endometrioma larger than 3 cm there is no evidence that cystectomy prior to treatment with ART improves pregnancy rates. In particular, The GDG recommends that clinicians counsel women with endometrioma regarding the risks of reduced ovarian function after surgery and the possible loss of the ovary. The decision to proceed with surgery should be considered carefully if the woman has had previous ovarian surgery.
Finally, the effectiveness of surgical excision of deep nodular lesions before treatment with ART in women with endometriosis-associated infertility is not well established with regard to reproductive outcome.
Iatrogenic menopausal symptoms in women with endometriosis
The GDG recommends that clinicians continue to treat women with a history of endometriosis after surgical menopause with combined oestrogen/progestagen or tibolone at least up to the age of natural menopause
Asymptomatic (incidentally diagnosed) endometriosis: what to do?
The GDG recommends that clinicians:
- Should not routinely perform surgical excision and ablation for an incidental finding of asymptomatic endometriosis at the time of surgery, since the natural course of the disease is not clear.
- Fully inform and counsel women about any incidental finding of endometriosis.
Primary prevention of endometriosis: fact or fancy?
According to GDG:
- The usefulness of oral contraceptives for the primary prevention of endometriosis is uncertain.
- The usefulness of physical exercise for the primary prevention of endometriosis is uncertain.
Endometriosis and cancer
The GDG recommends that clinicians inform women with endometriosis requesting information on their risk of developing cancer that: there is no evidence that endometriosis causes cancer and there is no increase in overall incidence of cancer in women with endometriosis.
Overview and conclusion
Interestingly, a substantial part of the recommendations is also similar to the previous one [5], indicating on the one hand a lack of recent, high quality studies in some areas and on the other hand similarities in retrieving the evidence from the literature by experts and the formal retrieving process of the evidence by a structured methodology of extensive literature searches. However, the main difference between the two guidelines is the structured methodology, based on the Manual for ESHRE Guideline Development, including objective assessment of the literature and an extensive and transparent review by relevant stakeholders.

Painting by Jodie Dunne, from Endometriosis Awareness through Art Association
References:
[1] Laganà AS, Sturlese E, Retto G, Sofo V, Triolo O. Interplay between Misplaced Müllerian-Derived Stem Cells and Peritoneal Immune Dysregulation in the Pathogenesis of Endometriosis. Obstet Gynecol Int. 2013;2013:527041.
[2] Eskenazi B,Warner ML. Epidemiology of endometriosis. Obstet Gynecol Clin North Am 1997;24:235–258.
[3] Meuleman C, D’Hoore A, Van Cleynenbreugel B, Tomassetti C, D’Hooghe T. Why we need international agreement on terms and definitions to assess clinical outcome after endometriosis surgery. Hum Reprod 2011;26:1598–1599; author reply 1599–1600.
[4] Dunselman GA, Vermeulen N, Becker C, Calhaz-Jorge C, D’Hooghe T, De Bie B, Heikinheimo O, Horne AW, Kiesel L, Nap A, Prentice A, Saridogan E, Soriano D, Nelen W. ESHRE guideline: management of women with endometriosis. Hum Reprod. 2014;29(3):400-12.
[5] Kennedy S, Bergqvist A, Chapron C, D’Hooghe T, Dunselman G, Greb R, Hummelshoj L, Prentice A, Saridogan E; ESHRE Special Interest Group for Endometriosis and Endometrium Guideline Development Group. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum Reprod. 2005;20(10):2698-704.




No Comments on Endometriosis: what’s the evidence for this enigmatic disease?
are you accepting referrals for excision of endometriosis?
4th June 2014 at 5:56 amOf course Nancy, I will be honored to accept referrals for excision of endometriosis from a high-skilled expert as you!
4th June 2014 at 7:09 pm