Assessing Composite Outcomes in Clinical Trials
Posted on 31st July 2020 by Yousif Eliya

“Name a ‘large’ RCT without a composite primary outcome?”
While you are looking for this ‘large’ trial, let’s set the objectives for this short blog:
- Introduce our case trial: HIP ATTACK from the Lancet
- Appraise the primary composite endpoint
- Verify our assessment
- Think of ways to interpret the result
PICO and Background
The HIP ATTACK investigators assessed whether accelerated surgery (defined as speedy surgery in less than 6 hours of diagnosis) compared to standard care in adult patients (45 years or older) with a hip fracture that required surgery, improved outcomes (1). A total of 2,970 patients were enrolled and the co-primary outcomes were death and a composite endpoint (CEP) of major complications (defined as mortality and non-fatal myocardial infarction (MI), stroke, venous thromboembolism (VTE), sepsis (defined with infection), pneumonia, life-threatening bleeding, and major bleeding) at 90 days.
Appraisal
The rationale behind the composite seems to be aligned with competing risk definition (i.e. there is a competing risk if the surgery results in the death of the individual, some of whom, if lived would experience an MI) (2).
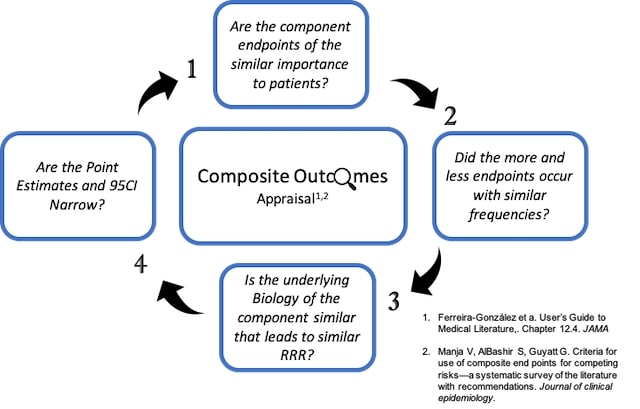
Are the component endpoints of similar importance to patients? Using Ferreira-González et al’s (3), classifications of this trial, the composite outcome are as follows: mortality (level I importance to the patient), non-fatal MI ( major III), stroke (possibly critical II), VTE (possible moderate IV), sepsis (possibly moderate IV), pneumonia (possibly moderate IV), life-threatening bleeding (possibly critical II), major bleeding (possibly major III). Hence, the gradient of the patient-importance is moderate.
Did the more and less endpoints occur with similar frequencies? Looking at Figure 1, the relative risk difference of mortality is 1%, 1% non-fatal MI, < 1% stroke, 0% VTE, 0% sepsis, 1% pneumonia, 0% life-threatening bleeding, and 1% major bleeding. The overall components occurred with closer frequencies. A component such as death, non-fatal MI, pneumonia and major bleeding occurred with equal frequencies.
Is the underlying biology of the component similar that it leads to a similar relative risk reduction (RRR)? Despite lacking clinical expertise, I think the biological rationale to have components such as non-fatal MI, pneumonia, and VTE to have a similar treatment effect is weak despite the intervention for this population (Figure 1).
Are the Point Estimates and 95% Confidence Interval (CI) narrow? The composite had an overall RRR of 17% (95 CI, -13% to 23%), critical/major patient-important components (death, MI, stroke) had point estimates (RRRs) of 9%, -5% and 65%, respectively. Similar to point estimate variability, the range of possible values that contain the true mean (95% CI) is also wide, ranging from 0.13 to 3.44 (Figure 1).
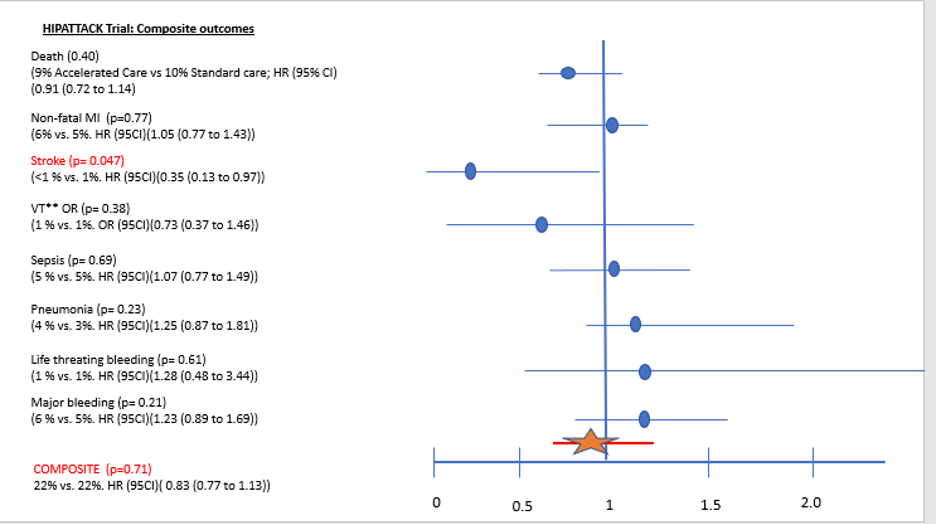
Figure 1
Assessment
The systematic assessment of the composite shows:
1) a moderate gradient of outcomes selected in the composite based on authors definitions;
2) overall closer frequencies among components with major CEPs having exact frequencies;
3) despite the weak biological judgment, RRRs differ widely, and 95%CI for stroke (critical outcome) is wide (0.13 to 0.97).
Therefore, point estimates and CIs for individual component endpoints should be reported to allow clinicians to make final decisions for their patients.
Interpretation
The authors of the HIP ATTACK trial reported the overall composite estimate in the abstract. Individual reporting of composite endpoints is also recommended based on our appraisal above. The authors could have reported their primary outcome as follows: the results of composite end-point (CEP) of major complications suggests that accelerated hip surgery, compared to standard care surgery, may reduce the risk for strokes but has uncertain effects on vascular death, non-fatal MI, VTE, sepsis, pneumonia, life-threatening bleeding, and major bleeding at 90 days follow-up.
Summary Figure
References
- Borges FK, Bhandari M, Guerra-Farfan E, Patel A, Sigamani A, Umer M, Tiboni ME, del Mar Villar-Casares M, Tandon V, Tomas-Hernandez J, Teixidor-Serra J. Accelerated surgery versus standard care in hip fracture (HIP ATTACK): an international, randomised, controlled trial. The Lancet. 2020 Feb;395(10225):698-708. doi: 10.1016/S0140-6736(20)30058-1.
- Manja V, AlBashir S, Guyatt G. Criteria for use of composite end points for competing risks—a systematic survey of the literature with recommendations. Journal of clinical epidemiology. 2017 Feb;82:4-11. doi: 10.1016/j.jclinepi.2016.12.001.
- Ferreira-González I, Permanyer-Miralda G, Domingo-Salvany A, Busse JW, Heels-Ansdell D, Montori VM, Akl EA, Bryant DM, Alonso-Coello P, Alonso J, Worster A. Problems with use of composite end points in cardiovascular trials: systematic review of randomised controlled trials. Bmj. 2007 Apr;334(7597):786. doi: 10.1136/bmj.39136.682083.AE.
Original cover photo by by Lea Böhm on Unsplash
You can read more about composite outcomes in this article on the Testing Treatments website.



