Catalogue of Bias: a resource review
Posted on 9th May 2019 by Sarah Tanveer

The first instance of a catalogue to organize biases found in health evidence was presented in David Sackett’s 1979 publication in the Journal of Chronic Disease [1] . He noted that biases may “distort the design, execution, analysis and interpretation of research” [1]. This catalogue was in part inspired by a graduate student at McMaster University, JoAnne Chiavetta, who noticed inconsistencies in research methodology used in clinical epidemiological research [2].
The Catalogue of Bias (CoB), is a digital resource for clinicians, students, researchers, investigators, and consumers of health evidence. As of posting this blog, the catalogue has 49 published biases, which also encompass the original 35 presented in Sackett’s (1979) paper:
How can you benefit from the Catalogue?
As a student, the catalogue is an invaluable reference tool not only to learn more about different types of biases, but also to avoid them in your own research. By having a catalogue of biases identified in one place, you can save both time and resources. Moreover, it may help reframe how clinicians, academics, and consumers apply evidence in their own decision making. I personally have referenced the catalogue when critiquing articles for journal club and when critically appraising the literature.
Catalogue Overview
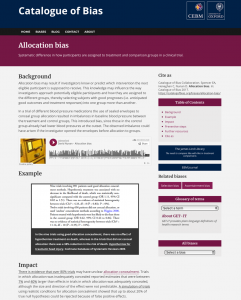
Each bias on the CoB website is organized in the following structure:
- Citation for the specific bias on the CoB
- Bias name and definition
- Background
- Example(s) of the bias in the literature
- Impact of bias
- Preventive steps
- Citations and/or sources
Once you open the bias of interest, there are several headers. To start, each bias is given a short definition that displays on the main catalogue screen.
The background section gives a brief overview of the bias selected, as well as any alternative names that may be used. For example, ‘verification bias’ can also be referred to as ‘work up bias’. It often also gives reasons for when these type of bias may occur.
The second section gives an example. It usually references a research publication or study that clearly demonstrates the bias.
The third section describes the implications or impact of the bias on the interpretation of results within the context of clinical practice. For example, the validity or accuracy of the findings may be influenced by the bias that was present in the research design.
The fourth section gives examples of preventive steps, or ways to mitigate these types of biases from impacting research. They offer suggestions on how to improve the methods used and, in some cases, recommend statistical corrections to help correct for bias. Additionally, at the end of each bias page are several sources and references for consumers to learn more about the bias.
Another very useful part of the catalogue is the glossary of terms; it provides easy to understand definitions of common terms used in EBM.
Future areas of improvement can be to create an interactive diagram that visually groups common types of biases. The CoB team is already working on creating a taxonomy which will organize the biases into different stages of study designs such as: design, setup, conduct, and reporting [3] , and their various interconnecting relationships.
Overall, the Catalogue of Bias offers researchers and consumers a succinct and helpful resource to reference when designing, analyzing, and interpreting findings from health research and papers. The catalogue is an evolving and living resource which will continue to develop over time. To send in suggestions for biases not already listed on the website, you can get in touch with the CoB editors [4] and join the conversation on Twitter using the hashtag #taxonomyofbias!