Comparing exercise in Parkinson’s Disease: The Berlin LSVT BIG Study
Posted on 22nd January 2014 by Lauren Smeltz

Ebersbach G, Ebersbach A, Edler D, et al. Comparing exercise in parkinson’s disease–the berlin LSVT BIG study. Mov Disord. 2010;25(12):1902-1908.
Parkinson’s disease (PD) affects the basal ganglia neurons that produce dopamine, a neurotransmitter that coordinates smooth muscle movements. Many individuals with Parkinson’s may exhibit tremors at rest, slowed movement referred to as bradykinesia, stiffness or rigidity, and/or balance difficulties or recurrent falls. For many this difficulty with movement can lead to difficulty performing activities of daily living such as, eating, dressing, as well as driving, walking, and getting around the community. There is no cure for Parkinson’s disease. Many may attempt to halt the progression of the disease with medications that act like dopamine in the brain. Even with pharmacological or surgical treatment many still exhibit movement difficulties. Exercise is an established therapeutic adjunct to other medical treatment and there are many studies that evaluate different techniques of therapeutic exercise.
I first encountered BIG therapy while I was on a clinical rotation. I had learned some of the principles of BIG therapy in class where we focused on encouraging patients with Parkinson’s to increase their amplitude by taking larger steps, trying to break the cycle of short shuffling gait patterns. Prior to being introduced to BIG therapy I had seen therapists use treadmill training as a means of increasing walking ability and speed in patients with Parkinson’s. This is how I developed the interest in which treatment was more effective at increasing independence in activities of daily living (ADLs) in Parkinson’s patients, BIG therapy or treadmill training? Once I began researching I found that there is not a lot of evidence out there that studies the effect of BIG therapy, or compares it to other interventions.
The study I found compared the effects of LSVT BIG therapy, Nordic walking (WALK) and unassisted home exercise (HOME). The authors do not give a detailed description of what LSVT BIG therapy is, they assume that if you are interested in this topic then you most likely know what the intervention entails. They do include a basic explanation of the principle and frequency of this type of intervention. They also do not describe Nordic walking, but by performing a Google search you can find images of what this intervention looks like.
The authors do a good job of expressing the inclusion and exclusion criteria. Patients were included if they had a Hoehn & Yahr stage of I-III. These stages represent individuals ranging from symptoms on one side only (unilateral) to balance impairment representing mild to moderate disease, but are physically independent. Individuals were receiving outpatient treatment and had to have stable medication 4 weeks prior to inclusion. They were excluded if they had dementia based on a mini-mental state exam of <25, severe depression, disabling dyskinesias (difficulty performing movement), or comorbidity affecting mobility or ability to exercise. These criteria represent the type of Parkinson’s patient you would typically see in an outpatient clinic, however they do not represent individuals with Parkinson’s that have a Hoehn & Yahr stage greater than III. 60 patients with Parkinson’s disease were referred from local outpatient clinics and office-based physicians that met these criteria and were randomly allocated to the intervention groups.

Parkinson’s community exercise group
The interventions were briefly described as I alluded to earlier. In order to reproduce this study it would be important to have a more detailed description of the exact exercises that were performed in the BIG group and the HOME group. Frequency and duration for BIG and Nordic walking were given, but they were not described for the HOME group. The frequency and duration for the BIG group is typical of a BIG program in the clinic. The HOME program would likely be similar to a community based Parkinson’s exercise group.
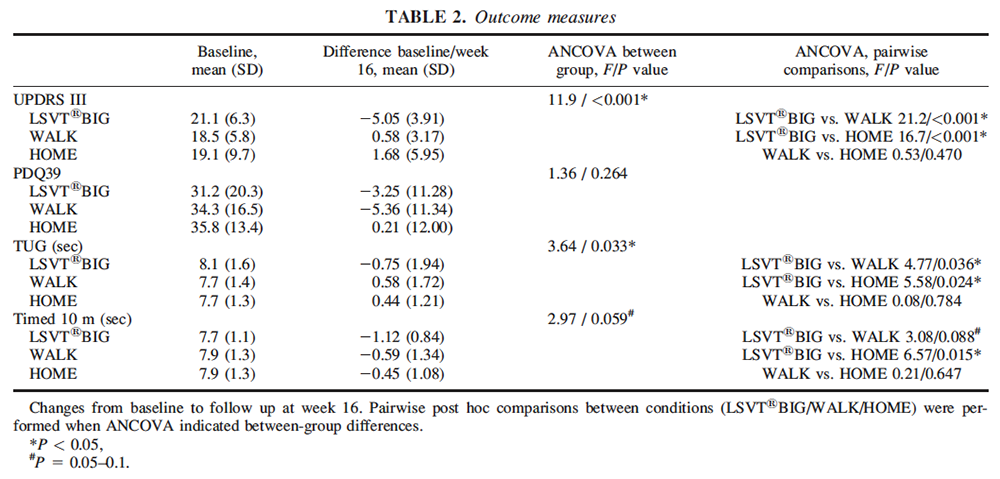
The outcomes that the researchers used were the unified Parkinson’s disease rating scale UPDRS section III (primary outcome), and secondary outcomes included the PD quality of life (PDQ-39), Timed Up-and-Go (TUG), and timed 10 meter walk. Individuals were video taped performing the UPDRS and then rated by an experience rater so that the rater was blinded for group allocation and time-point of examination. These assessments compare measurable and functional outcomes as well as quality of life. All assessments were taken at baseline and at 16 weeks from baseline measurement. Therefore this study shows whether outcomes were maintained even after the intervention was terminated. Frequently, therapists are unaware of whether patients maintain their physical gains after cessation of treatment and it is important that an intervention can produce sustainable results.
Differences in change from baseline to week 16 between groups were assessed using analysis of covariance (ANCOVA), which is a measure of how much two variables change together while statistically controlling for the variable that is not of primary interest (covariate), in this study the covariate was the baseline values. If there was a significant overall difference between groups, then pairwise comparisons were performed. ANCOVA showed significant differences for UDPRS-motor scale at final assessment. When compared pair wise, LSVT BIG was superior to WALK and HOME. Also there was a difference for TUG and pairwise comparison showed a better outcome in LSVT BIG compared to WALK and HOME. There was a tendency for group differences for timed walking with a better outcome in LSVT BIG compared to WALK and HOME. There were no significant differences for PDQ-39. There were no pairwise differences between WALK and HOME for any of the outcome measures. Table 2 (below) does an excellent job of depicting the changes in measures from baseline to week 16 and they are fairly easy to understand even with very little knowledge of statistical significance.
This study shows significant justification for the use of LSVT BIG therapy in patients with PD of mild to moderate severity when compared to these other 2 specific interventions. The mean improvement on the UDPRS of 5.05 points is nearly two times the minimal clinically important difference (MCID)–statistical model to identify the smallest change in a treatment outcome that a patient would identify as important–which in itself is pretty powerful in justifying the quality of LSVT BIG as an intervention for PD. Because there is no cure for Parkinson’s disease it is important that we find more evidence to support interventions that are successful at slowing the progression of the disease. More research needs to be done to compare this to other types of one to one treatment in the clinic. Also, LSVT BIG therapy as performed here and as is recommended at 1 hour per day and 4 days per week is very time consuming. It would be interesting to see if LSVT BIG therapy could have similar outcomes when performed 2-3 times in the clinic combined with a home program because this is more typical of what treatment schedules available in the clinic and more typical of what some types of insurance are willing to reimburse.



No Comments on Comparing exercise in Parkinson’s Disease: The Berlin LSVT BIG Study
Thanks. As the partner, and researcher, of someone with PD your intelligent and impartial assessment is really appreciated.
12th December 2019 at 6:33 amTratamento possui uma vez que principal ação fomento inquieto que faz com
15th October 2016 at 8:39 amque nossa circulação sanguínea seja estimulada, além de
uma maior oxigenação celular.
This fact might partially clarify the inconsistency of needling depth of every single acupuncture
12th August 2016 at 3:22 ampoint as reviewed in our study.