Treatments can harm
Posted on 12th June 2017 by John Castle

This is the first in a series of 36 blogs based on a list of ‘Key Concepts’ developed by an Informed Health Choices project team. Each blog will explain one Key Concept that we need to understand to be able to assess treatment claims.
This is one of the first and foremost reasons that we need to test treatments. Medical Ethics principle number 2: Do No Harm.
It may seem like common sense, but many examples throughout history have shown that, without an evidence-based approach, people can be harmed or even killed by a treatment. Few effective treatments are 100% safe and industries will time and again downplay potential harms and exaggerate benefits. Often the treatment hasn’t been tested adequately or data concerning adverse effects has not been properly recorded or presented.
Example 1. Antiarrhythmic drugs in patients having a heart attack
After having a heart attack, some people develop heart rhythm abnormalities – arrhythmias. Those who develop arrhythmias are at higher risk of death than those who don’t. Since there are drugs that suppress these arrhythmias, it seemed logical to suppose that these drugs would also reduce the risk of dying after a heart attack.
The drugs had been tested in clinical trials, but only to see whether they reduced heart rhythm abnormalities and not whether they would reduce the risk of dying after a heart attack. When the accumulated evidence from trials was first reviewed systematically in 1983, there was no evidence that these drugs reduced death rates [1]. In fact, the drugs had exactly the opposite effect. However, they continued to be used – and continued to kill people – for nearly a decade.
At the peak of their use in the late 1980s, one estimate is that they caused tens of thousands of premature deaths every year in the USA alone. In fact, they were killing more Americans every year than had been killed in action during the whole of the Vietnam war [2].
This example illustrates how analysing the wrong outcome measure can be misleading.
Example 2. Dr. Spock
Advice can also be lethal. Many people have heard of the American Childcare specialist Dr. Benjamin Spock, whose best-selling book Baby and Child Care became a bible for both professionals and parents, especially in the USA and the UK, over several decades. However, in giving one of his pieces of well-meaning advice, Dr. Spock got things badly wrong.
With a seemingly logical suggestion, he said: ‘There are two disadvantages to a baby sleeping on his back. If he vomits he’s more likely to choke on the vomitus. Also he tends to keep his head turned towards the same side… this may flatten the side of the head… I think it is preferable to accustom a baby to sleeping on his stomach from the start’.
This piece of advice became common practice despite not being rigorously evaluated, and it is estimated to have caused tens of thousands of avoidable cot deaths [3].
When clear evidence for these harmful effects emerged, the practice of letting babies sleep on their fronts was abandoned and cot death numbers started to fall dramatically.
This shows how expert opinion is not always correct. (This is discussed further in a later blog in this series. Key Concept 1.6: expert opinion is not always right).
Example 3. Thalidomide (Sorry, but it’s really important)
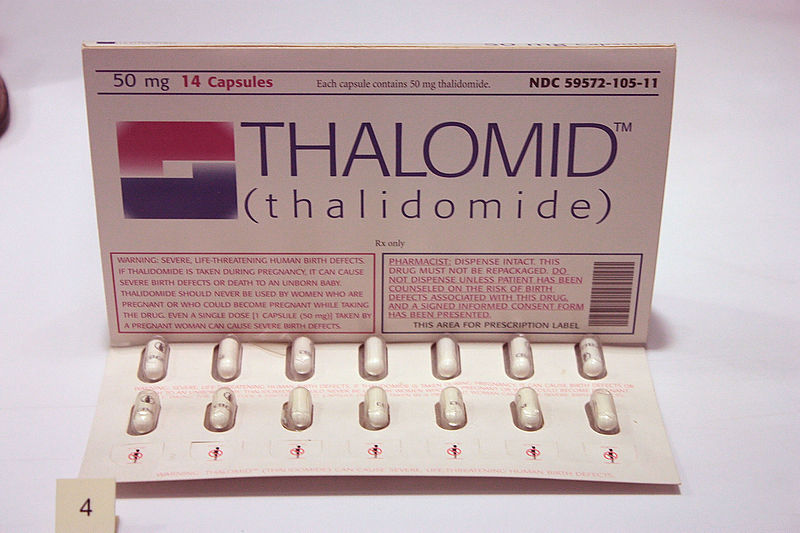
Thalidomide is an especially chilling example of a new medical treatment that did more harm than good because it wasn’t tested for its purpose. This sleeping pill was introduced in the late 1950s as an apparently safer alternative to the barbiturates that were regularly prescribed at that time. Unlike barbiturates, overdoses of thalidomide did not lead to coma. Thalidomide was especially recommended for pregnant women in whom it was used to relieve morning sickness.
Then, at the beginning of the 1960s, obstetricians began to see a sharp increase in cases of severely malformed arms and legs in newborn babies. This previously rare condition results in such extremely shortened limbs that the hands and feet seem to arise directly from the body.
Doctors in Germany and Australia later linked these infant malformations with the fact that the mothers had taken thalidomide in early pregnancy [4]. Resulting in patient monetary claims amounting to millions of pounds.
The thalidomide tragedy stunned doctors, the pharmaceutical industry, and patients, and led to a worldwide overhaul process of drug development [5].
Examples such as these are more numerous than you’d care to imagine, and they all result from practitioners, researchers and pharmaceutical companies not testing the treatment properly – resulting in the harm or death of thousands of people and the unnecessary expense of hundreds of healthcare bodies.
So, ultimately, we need evidence because we need to know if a practice does more harm than good and we should always consider the possibility that treatments can harm.
Some content republished from Testing Treatments with permission.




No Comments on Treatments can harm
Excellent! I think these examples will do way more to help people understand EBM than anything else. EBM evolved because our way of treatment was not working and killing thousands!
18th July 2019 at 12:55 pmWell done, John. A nice post to begin the series.
13th June 2017 at 2:17 pm