Cochrane Crowd: Harnessing the Power of the Crowd in Evidence-Based Healthcare
Posted on 8th July 2016 by Pishoy Gouda

As of 2015, there have been over 200,000 clinical trials registered around the world. These trials have created a tremendous amount of data to help answer important everyday clinical questions. Should this patient with a heart attack go for a triple bypass? Does a new cancer medication work? Is this patient suitable for a c-section?
These are the types of questions that trials can help answer. Unfortunately, different trials don’t always agree with each other and there are a lot of reasons why this might be. Sometimes there are flaws in the study design, differences in study populations or it could be just due to chance.
Systematic reviews such as Cochrane Reviews often help us definitively answer these important questions, by synthesising and analysing all the available trials for a specific clinical question, such as ‘which patients with a heart attack (STEMIs) should undergo invasive management?’ – important questions for which clinicians need answers.
When we look at individual studies we don’t always get the full picture. Systematic reviews aim to help overcome this by collecting all the relevant studies that can help us answer the question. A key part of the process is usually a search of scientific electronic databases, such as PubMed. However, such searches can retrieve an extraordinary number of hits. When Fanning et al. conducted a search to answer this very question they had to go through over 2000 articles to find the 8 that were ultimately important to them.
The problem
With the plethora of articles being published everyday, it is becoming increasingly difficult for researchers to identify studies that are relevant to their clinical question.
This leads to potential delays in the production of reviews that shape how we practice medicine. This delay is not just a theoretical problem; it has real consequences. Every day that we delay changing our clinical practice, in the presence of suitable evidence, patients and their families suffer the consequences.
Keeping up with this information overload has become a very real and significant challenge that we need to face.
A solution: Cochrane Crowd
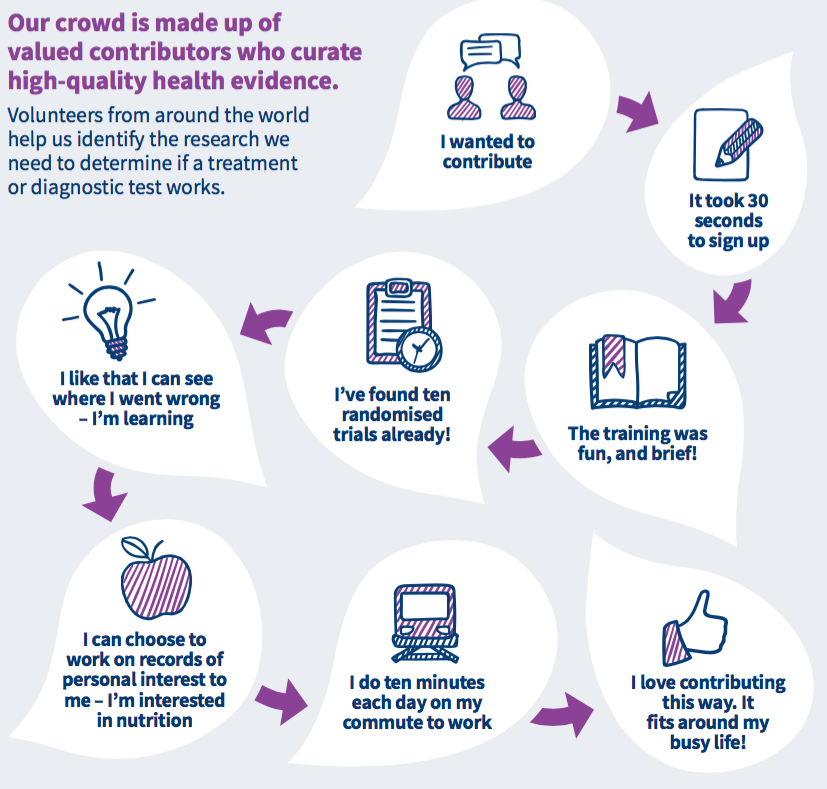
This is where you come in: become a Cochrane citizen scientist! Cochrane has recently launched a new platform called Cochrane Crowd and, building on the previously very successful Embase screening project, this platform hosts micro-tasks that are aimed at tackling the data deluge! Contributors help categorise and summarise healthcare research evidence. Currently, nearly 3000 people have signed up to help, from 31 different countries, and almost a million classifications have been made. No previous experience is necessary and it only takes 30 seconds to sign up.
You’ll be welcomed with the following: “Your mission, should you choose to accept it, is to help us identify reports of randomised controlled trials (RCTs)”.
You don’t even need to be familiar with RCTs, as you will be be guided through the task.
If you choose to accept this mission, you can work at your own pace, whenever you want, and just a few minutes a day can make a big difference. Essentially, you work your way through citations (as many or as few as you like at any time) to identify which of them are describing randomised trials and which aren’t. You can also choose to screen particular citations of interest to you (e.g. you can screen citations relating to ‘depression’ or ‘stroke’). You can track your progress and learn as you go because you can see where the final decision about a citation did not agree with the classification you gave it.
The citations you screen feed the Cochrane Central Register of Controlled Trials (CENTRAL). This register is a vital resource for systematic reviewers and other researchers trying to find the evidence about a treatment or intervention. Helping to make this resource as up to date and as comprehensive as possible is really important.
So if you are ready to help make a difference and shape how we practice healthcare, check out Cochrane Crowd: http://crowd.cochrane.org
If you have more questions, see the Frequently Asked Questions for Cochrane Crowd or take a look at the infographics below.